Medical Delivery Technology
Developing medical-grade delivery systems for cannabinoid-based medicines. Built for dosing accuracy, adherence, and real-world data—designed for clinics and pharmacies, not the recreational market.
Engineered for Clinical Reality
Most cannabis hardware on the market today was never designed for medical use. Nooetic Devices takes a strictly clinical route. We do not chase consumer trends; we engineer for stability, safety, and repeatability.
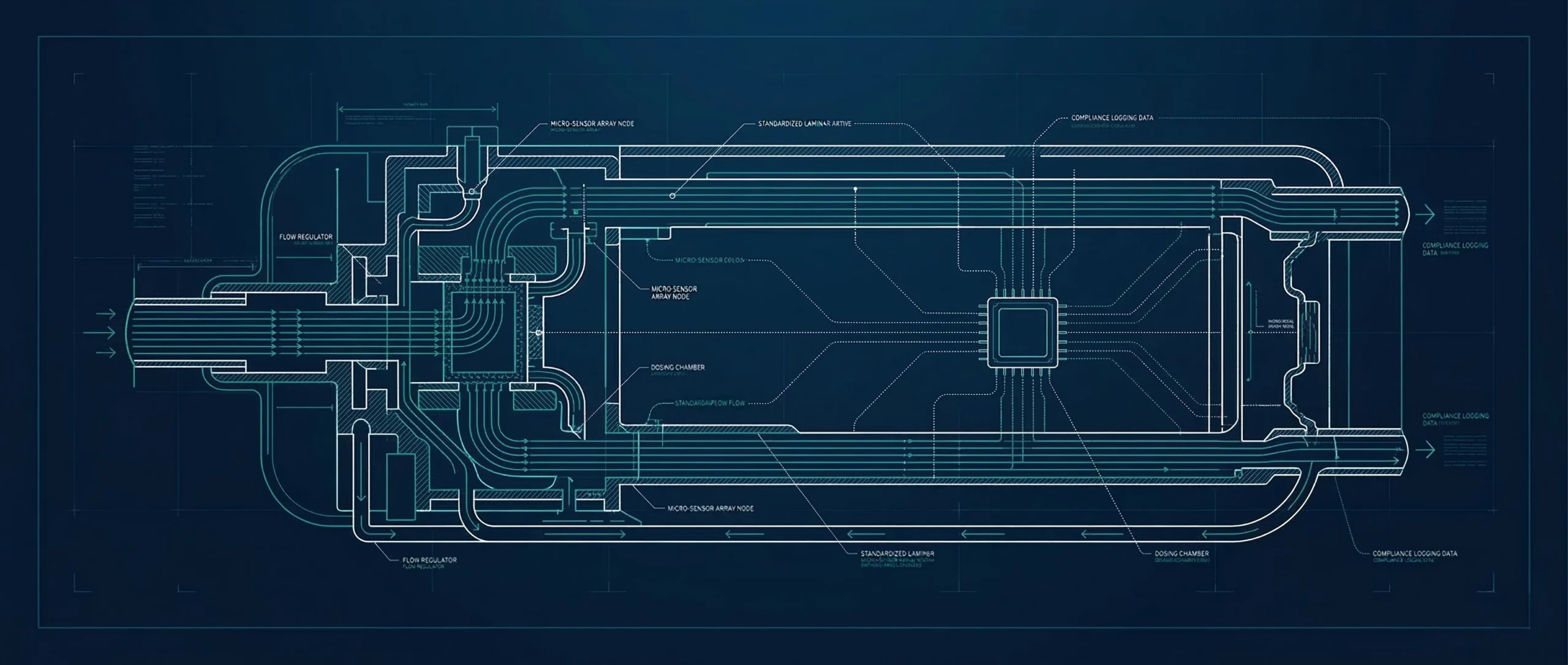
Proven Platforms
Starting with established, auditable device architectures rather than unverified consumer hardware.
Clinical Refinement
Adapting and refining hardware strictly for regulated medical environments and licensed pharmacies.
Data-Driven Design
Engineering around precision dosing mechanics, secure data capture, and trackable patient adherence.
Regulation & Data
Devices do not exist in a vacuum. They must function securely within regulated medical systems and clinical pathways.
Clinical Alignment
Aligning device usage strictly with established prescribing pathways in Nooetic Clinic and our broader partner networks.
Evidence Generation
Prioritizing real-world data capture and adherence monitoring to support medical positioning and market access.
Regulatory Sequencing
Working closely with manufacturing partners to define realistic, step-by-step paths for device adoption across legal jurisdictions.
The Integrated Advantage
We give partners the opportunity to link Product + Device + Clinical Pathway, eliminating the friction of managing three separate supply chains.
Nooetic Wholesale
Provides access to a broad range of compliant, EU-GMP cannabinoid products specifically engineered to be paired with our device platforms.
Nooetic Clinic
Creates real-world clinical environments where delivery systems can be utilized, evaluated, and refined in line with actual patient and prescriber needs.
Nooetic Bio Tech Group
Coordinates the commercial architecture, ensuring hardware development is tied to actual market demand and regulatory compliance.
Collaboration & Development
We are open to exploring structural partnerships that reflect shared development, IP contributions, and long-term commercial rollout plans.
Device OEMs
We partner with original equipment manufacturers looking to adapt and certify existing hardware platforms for regulated cannabinoid use.
Software & Tech
Collaborating on software infrastructure for real-world data capture, patient adherence tracking, and clinical monitoring solutions.
Clinics & Pharmacies
Supporting healthcare groups interested in rolling out consistent, standardized device platforms backed by a compliant product supply chain.
Brand Owners
Structuring co-branded or white-label medical devices strictly aligned to a stable, traceable medical supply chain.
Let’s Talk Medical Hardware.
If you are developing hardware, running clinics, or building a medical brand and want to explore delivery systems that actually fit regulated environments, we are ready to talk.
Discuss Device Collaboration →
