Quality & Compliance
Regulation isn’t a box to tick at the end—it’s how we design everything from day one. If it doesn’t stand up in front of a regulator, an auditor, or an institutional partner, we are not interested.
The Rules That Matter
Depending on the activity and jurisdiction, our structures and partners operate within strict, verifiable frameworks. We work exclusively with entities holding the appropriate licenses for their part of the chain.
Clinical Governance
Operating under strict clinical regulation, including the Care Quality Commission (CQC) and equivalent national healthcare bodies.
Controlled Drugs & Medicines
Strict adherence to medicines and controlled drugs legislation via appropriately licensed pharmacy and distribution partners.
Production Standards
Cultivation, production, and processing executed strictly under recognized GMP and EU-GMP standards.
Logistics & Data
Fully licensed import/export control for cross-border movement, backed by uncompromising UK/EU GDPR data protection obligations.
Documented & Repeatable
Good governance isn’t just policies on a shelf—it’s how decisions actually get made. This gives partners and investors absolute confidence that our theoretical standards match our operational practice.
Standard Operating Procedures
Strict, written SOPs governing clinical workflows, supply chain handling, data security flows, and customer service.
Incident Management
Clear, documented escalation routes and reporting paths for operational issues and clinical incidents.
Continuous Audit
Ongoing operational review and infrastructural improvement as markets, clinical regulations, and partners evolve.
Data Handled the Right Way
We use strict data processing and sharing agreements so every party’s responsibilities under GDPR are impeccably defined. We are highly defensive regarding who holds data and why.
Nooetic Bio Med Group
- Controls marketing and referral data architecture
- Manages commercial CRM systems
- Oversees B2B partnership information
- No direct clinical data access
Clinical Partners / Clinics
- Sole controllers of patient and medical data
- Manage all secure clinical records
- Control prescribing and follow-up data
- HIPAA/GDPR compliant medical storage
Third-Party Agencies
- Operate exclusively under strict written agreements
- Act on Nooetic’s instructions only
- Do not own or silo patient data
- Zero ability to make clinical decisions
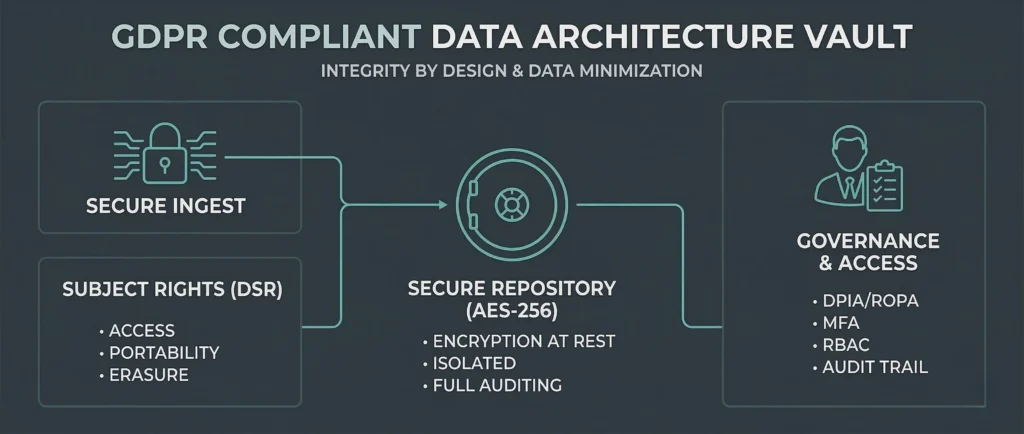
Traceable by Design
We would rather walk away from a deal than build on a supply route that cannot be defended. Our partners have a transparent understanding of where products are made, how they are processed, and how they move.
- Producers operating under verified GMP/EU-GMP
- Batch-level traceability from cultivation to pharmacy
- Immediate access to full Certificates of Analysis (COAs)
Clear Accountability
The goal is not to eliminate all risk—that is impossible in emerging markets—but to understand it, share it sensibly, and manage it securely through structured contracts.
- Clearly defined responsibilities for applicable regulations
- Established Joint Governance Committees
- Contractual clarity on regulatory breaches and corrective actions
Verify Our Compliance Framework
If you are a clinic director, pharmacy, manufacturer, or institutional investor and want to dig deeper into how our quality processes are architected, we are ready to open the books.
Request a Compliance Overview Call →
